Validation Study: Basic overview
This Article is written here to have an overview on the validation Study, now a day’s validation became an integrated part of cGMP. As we all know that the pharmaceutical industries are manufacturing the life saving drugs, hence the key objective set by the pharmaceutical industry is to manufacture products of required attributes and quality with consistency, at the lowest conceivable cost.
Validation study is the first requirement of any regulatory authority to give approval of commercialization / Market authorization. Various regulatory authorities defined the guidance on the Validation study and its approach for execution; here in this article we are going to have an overview on complete validation study.
Inside Story
- What is validation study?
- History of Validation.
- Scope (Phases) of validation.
What is Validation study?
History of validation study
Concept of validation is primarily derived from US FDA (Food and drug association) regulations describing the current good manufacturing practice (cGMP) for finished pharmaceuticals (21 CFR parts 210 and 211).
USFDA was the pioneer to advocating the concept of validation. In the mid of 1970’s this concept was proposed by two FDA officials names Ted Byers and Bud Loftus. This concept was introduced looking at the several problems in the sterility of large volume parental Market. First draft guidance on General principles of Process validation was come in May 1987.
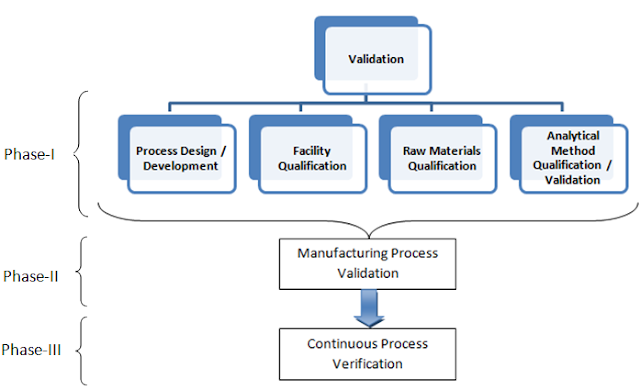
As we defined, in the above section the term validation is the process to prove operational and mechanical consistency, scope of validation study program is very vast, it covers every aspect of the pharmaceutical industry from its establishment to commercialization of product. An organized overview on validation study scope defined here, which involves three phases of validation;
- Phase-I: Pre-validation / Qualification Phase (Prerequisites of Process validation)
- Phase-II: Validation Execution Phase (Process Validation)
- Phase-III: Validation Maintenance Phase (Continuous process verification)
- Process Design / Development: This Study is comprise of various Process Understanding Studies which includes; Process control study, Hold time study, In-process control study, Filtration study, Drying Study, Shipping validation, Process Data Trending etc.
- Facility Qualification: This study is comprised of the study on various parts of a manufacturing facility which allows manufacturing of product within; likewise Utility Qualification, Equipment Qualification and calibration study program, Equipment Cleaning Validation, Operator Qualification etc.
- Raw Materials Qualification: Raw material Qualification study comprises of Raw material Analysis, Vendor/supplier Qualification and Transportation validation study etc.
- Analytical Method Qualification / Validation: Analytical Method Qualification study involves the qualification study for the analytical methods Residuals Assay Qualification, In-process Method Validation, Release Method Validation etc.
Phase-II: Validation Execution Phase (Process Validation)
In the first phase of validation i.e. Pre-validation / Qualification Phase we have understood the things need to be established prior to validate a manufacturing process of a drug substance (API) / Drug Product. This study is designed to validate or authenticate that an established sequence of process, established acceptance criteria and critical / non critical process parameters are suitable and adequate.
This study is a type of declaration that particular process can produce with the consistency operation / procedural and quality point of view over the period of time even under the worst conditions.
Phase-III: Validation Maintenance Phase (Continuous process verification)
This is the maintenance phase of manufacturing process of drug substance (API) / Drug Product.
As the name suggest it requires a frequent review of all process/product related documents which involves Manufacturing process, Quality management system (QMS) documents, analytical documents etc. Intention for the review of all this documents is to ensure that it have been no changes have been made / occurred in the process after execution of Phase-II i.e. process validation study, No Failure, deviations and modification took place without any investigation or acknowledgement.
This maintenance study could be an alert system design and validation of the system that shows high degree of assurance that all the lots and batches manufacture will achieve their particular established specifications.
I hope this article on basic introductory knowledge on validation study in pharmaceutical industry, helps you to boost up your knowledge stock of your mind.
Other useful articles may informative…..
- Product Lifecycle Management: An Overview
- Pharmaceutical regulatory authorities All over the world
- Pharmaceutical Quality Management System: An overview
- What is Quality Management system for pharmaceutical industry?
- Elements of Quality management system: An overview
- Pharmaceutical Drug substance (API) and Drug Product: Definition
- Research and Development: Definition













2 Comments
Very informative and nicely presented.
ReplyDeleteThank you sir....!!
DeleteIf you have any doubts, Please contact us